
Canadian Public Reimbursement Timelines
Improving access to new medicines is a critical challenge within Canada’s healthcare system – and one of Innovative Medicines Canada’s key focus areas.
We believe that transparent information-sharing is essential to collaboratively identifying solutions and strengthening the sustainability of Canadian health systems. To support this effort, Innovative Medicines Canada has created this resource hub to provide updates and trends related to drug reimbursement timelines in Canada and OECD countries.
This page is regularly updated with data and analysis on public reimbursement processes, including:
- Time to access new medicines
- Volume of negotiations underway
- Key pCPA performance metrics
The data is independently collected and aggregated by IMC, using sources such as IQVIA International Reimbursement Comparison, Health Canada, Canada’s Drug Agency (CDA), Institut national d’excellence en santé et services sociaux (INESSS), pan-Canadian Pharmaceutical Alliance (pCPA), and various Canadian provincial formularies.
CANADIAN PUBLIC REIMBURSEMENT TIMELINES RESOURCES
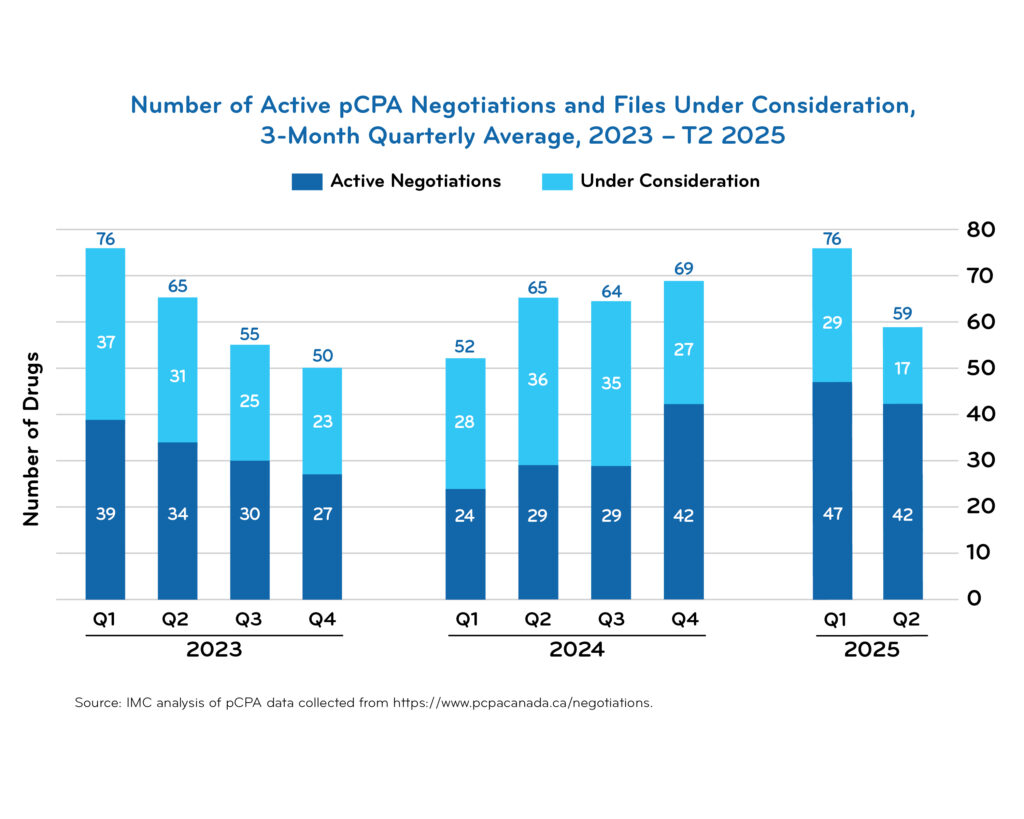
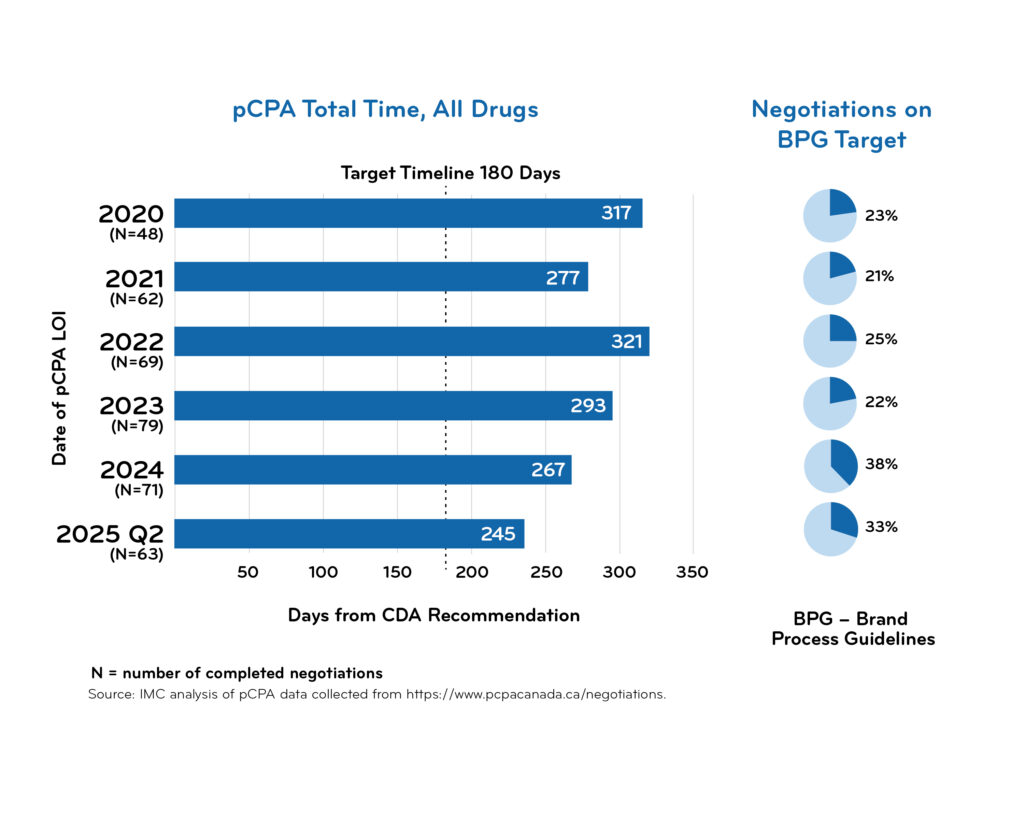
pCPA timeline data
Access downloadable trend charts reflecting the most recent quarterly data:
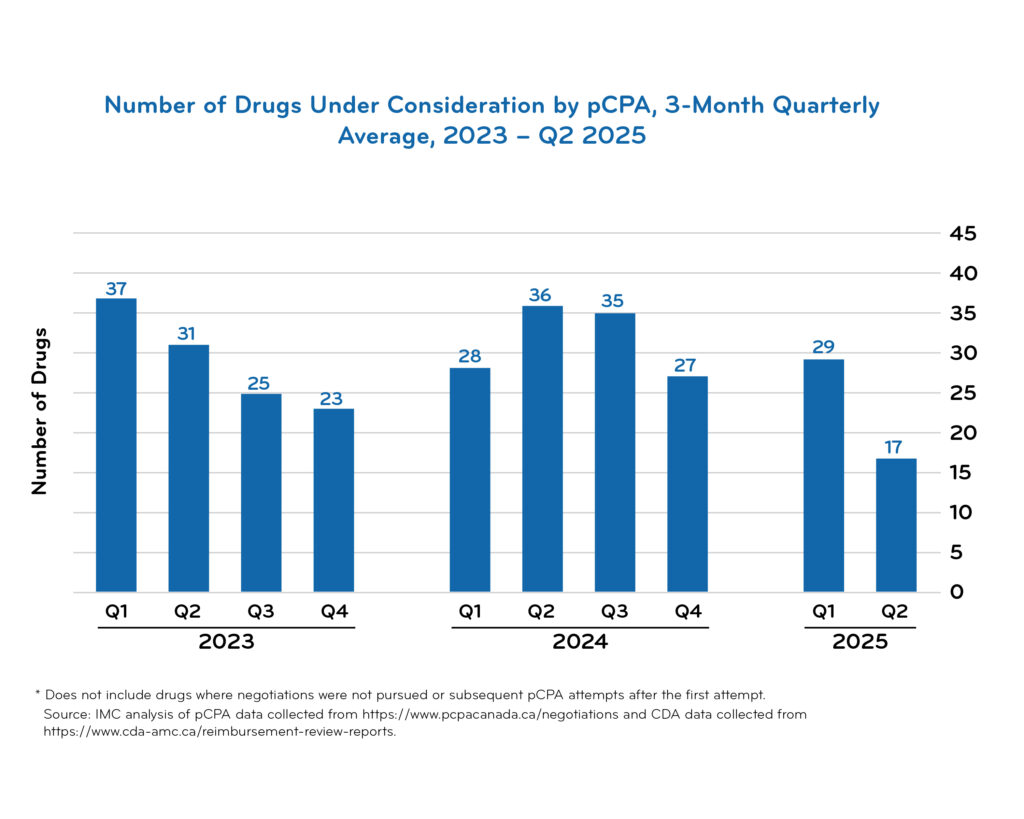
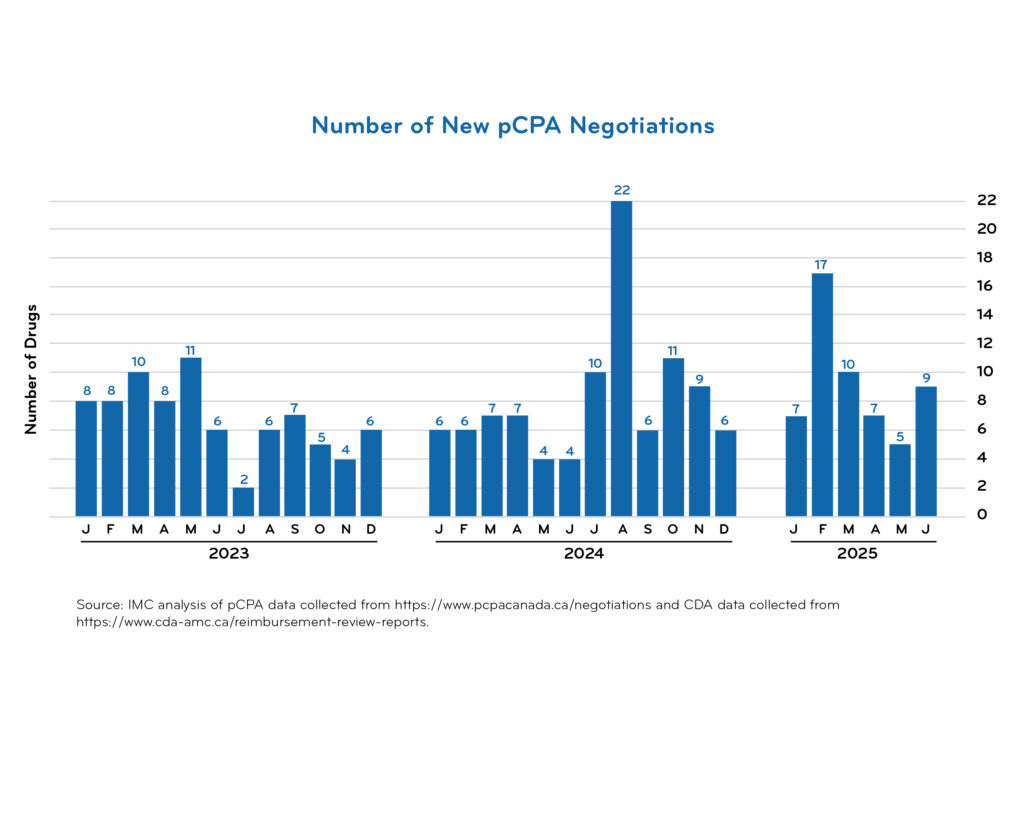
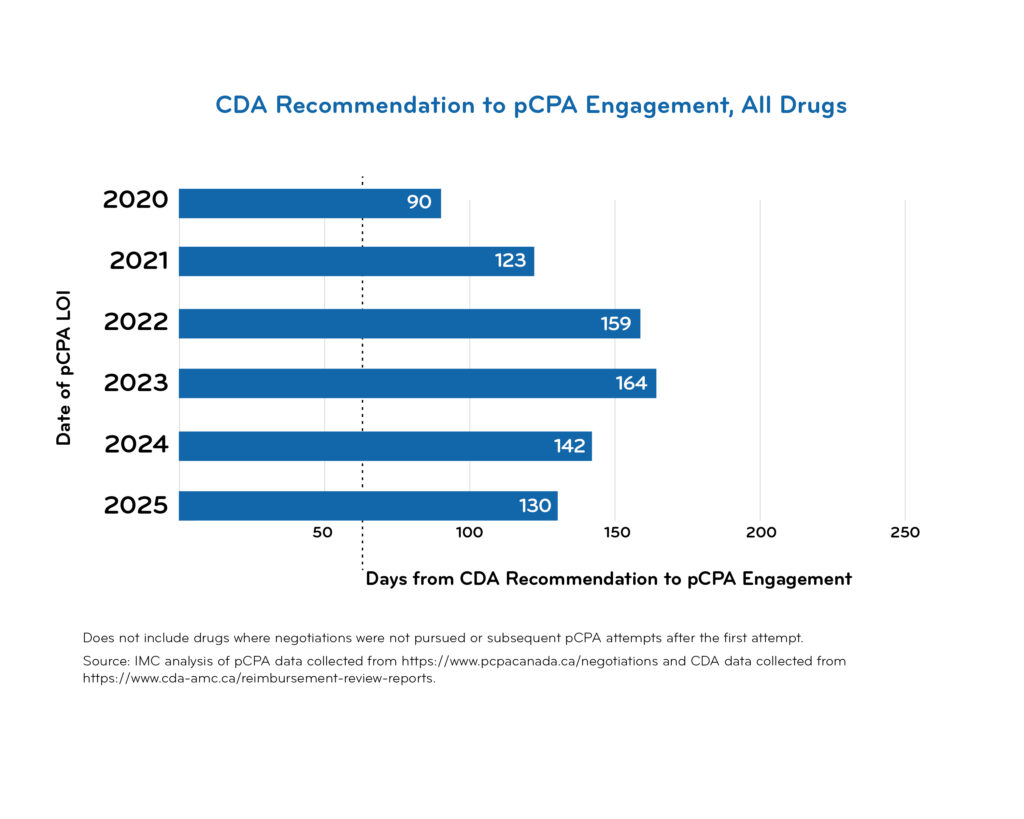
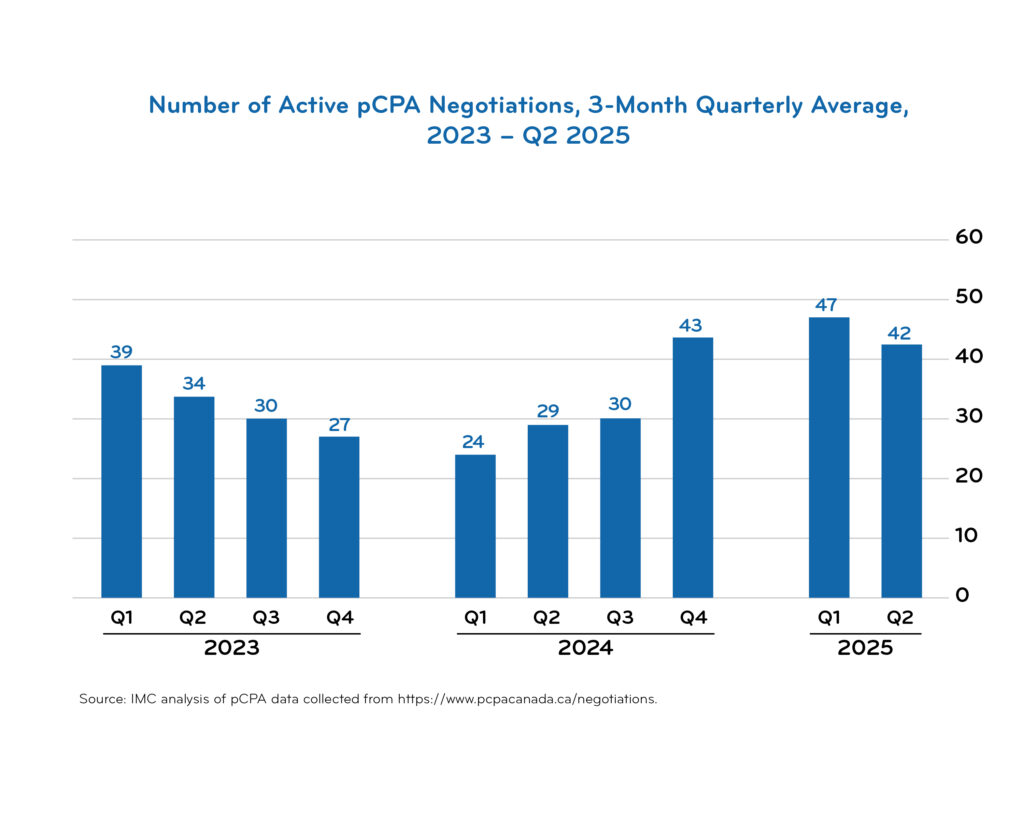
Additional pCPA resources

Innovative Agreements Framework
Many products coming to market are poorly served by traditional negotiation approaches. This document describes innovative agreements being used in jurisdictions outside Canada. We prepared it to guide discussions with the pCPA Governing Council on the merits of jointly developing an innovative agreements framework.
Additional analysis
- MORSE Consulting’s review of the latest trends and insights
- MORSE Consulting’s 4th Annual pCPA Year in Review Infographic (2020 Edition)
- MORSE Consulting pCPA February 2021 Trends and Insights
These third-party resources offer additional perspectives on drug reimbursement timelines and policy trends in Canada.



